|
In addition, hydrogen has at least one “active” lone pair. Conversely, these atoms have a high negative charge. Hydrogen attaches to highly electronegative atoms and acquires a high positive charge. They form due to the unequal charge distribution in the molecule. It would help if you remembered that hydrogen bonds do not form spontaneously. Therefore, hydrogen bonds are an essential aspect of chemical bonding. The greater the hydrogen-atom-atom ratio, the stronger the hydrogen bonds will be. As you go down the group, the boiling point of the compound increases. The PH3 molecule has a low boiling point, and the hydrogen atom is attached to one of the electronegative atoms. Instead, the London dispersion forces occur between the nh3 molecules. NH3 can form hydrogen bonds with water, but PH3 cannot. The molecule does not have a trigonal planar geometry and a dipole moment of 0.58D. PH3 is a polar molecule with a lone pair on one atom. However, ch3nh2 or ch3oh can form hydrogen bonds between molecules of the same type. Moreover, phosphorus cannot be used as a proton acceptor, as nh3 is a nearly universal proton acceptor. 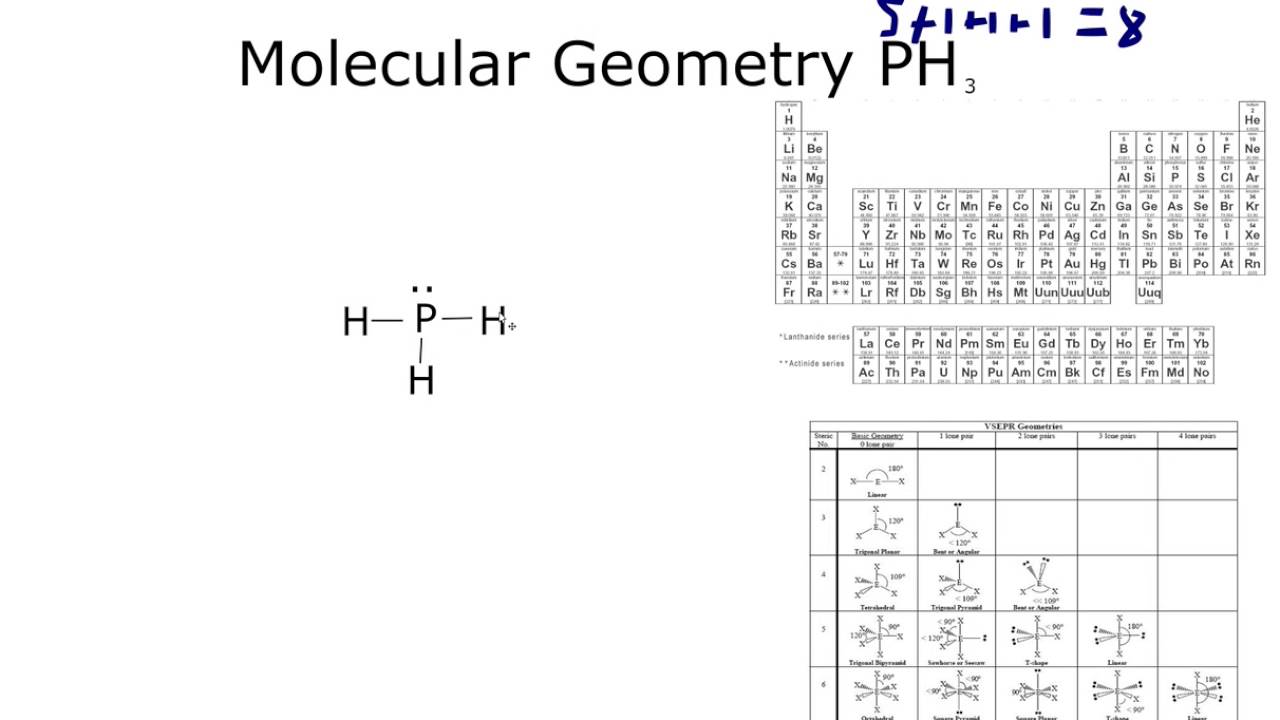
In addition, the phosphorous atom’s electrons are located in the third orbital, far from the nucleus. This is because it cannot render the opposite charge on the hydrogen-bonded. The phosphorus atom is a poor candidate for hydrogen bonding. These interactions are responsible for deviations from the ideal gas law. The electrostatic forces fall off with increasing distance between two molecules, and these interactions become essential at high pressures. These interactions are a sum of both repulsive and attractive forces. These interactions occur when positive or negatively charged species interact with one another. Hydrogen-hydrogen bonds are also a result of electrostatic interactions between two molecules. Hydrogen bonds also play a vital role in a molecule’s nucleotide bases. The two molecules then form a dipole-dipole intermolecular force, which requires considerable energy to break. The partially positive end of the hydrogen atom is attracted to the partially negative end of the oxygen atom. Hydrogen bonds occur when the hydrogen atom is bonded to oxygen, nitrogen, or fluorine. Moreover, the PH3 dipole-dipole interactions also have a pronounced effect on the boiling and melting points of the substance. 
However, because hydrogen bonds are more robust than dipole-dipole interactions, they can be used to separate polar molecules in solution. These interactions are weaker than the dipole-dipole interactions of most other molecules, including water. Hydrogen-hydrogen interactions only happen between organic molecules, forming hydrogen bonds between them. The phosphine molecules have a dipole moment of 0.58D, much smaller than the NH3 dipole moment. These forces are more potent than the Van der Waals forces. The molecules of the interhalogen compound PH3 form a dipole-dipole interaction and a hydrogen bond. A molecule with a similar structure to a PH3 molecule is a tetrahedral molecule. A polar molecule, such as carbon dioxide, is a tetrahedral molecule. The lone pair on the central O contributes to its polarity. However, its planar shape makes it nonplanar. Therefore, a molecule is polar if its electronegativity is less than 0.4.īecause of the P-H bond, PH3 is a polar atom. 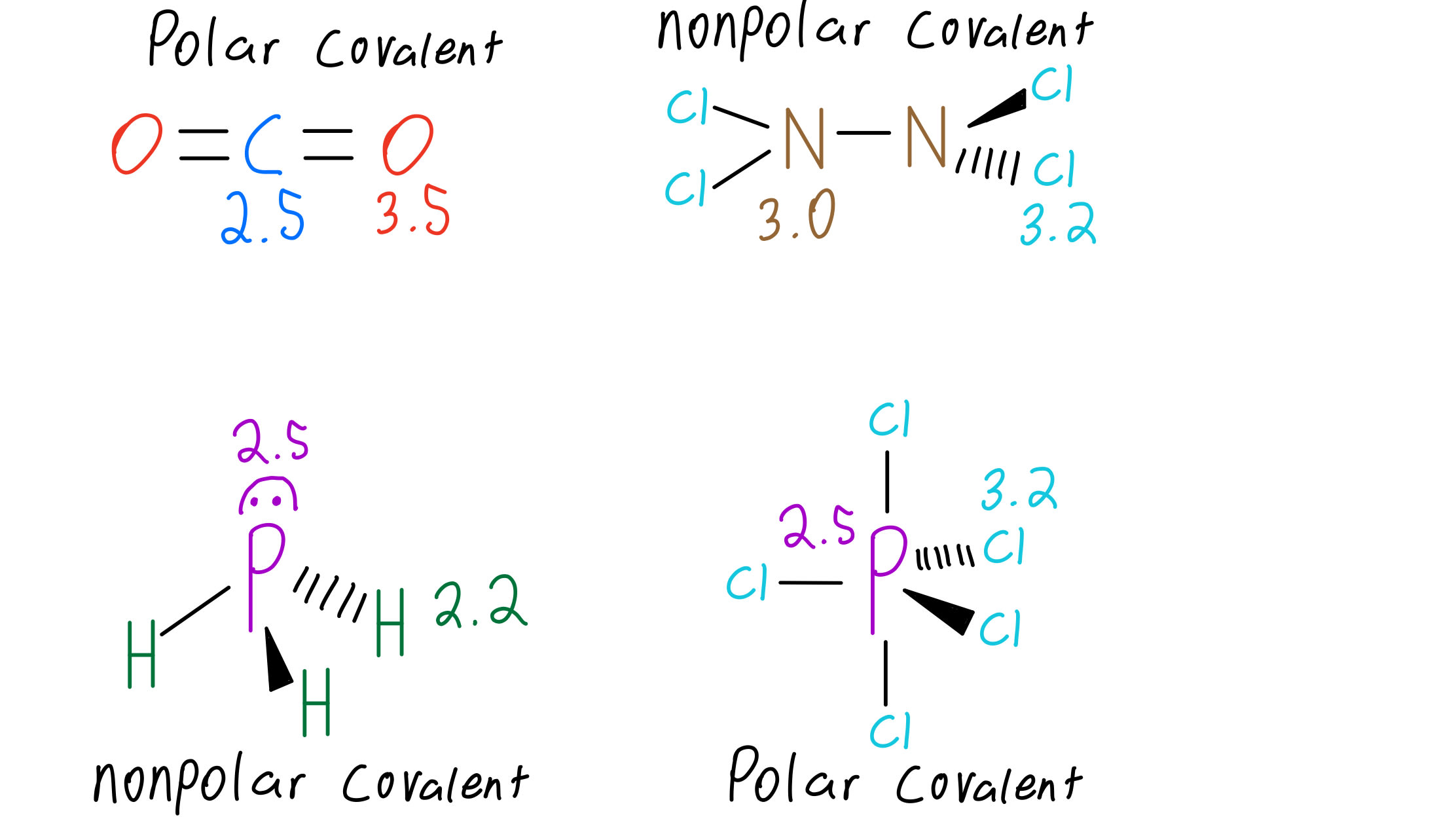
The two types of molecules share electrons symmetrically. Nonpolar molecules contain one type of atom, while symmetrical molecules contain two or more atoms. The electronegativity of a molecule varies widely, and the lone pairs on outer atoms are considered nonpolar. Hence, the polar molecule is a water molecule. Therefore the lone pair pushes down when compared to the lone pair. Unlike hydrogen, which only needs two valence electrons to form a fuller outer shell, phosphorous has a dipole moment that is less than one D. 
Though $P$.PH3 has a Lewis structure that contains eight valence electrons. Hint: The size of the P atom is greater than the N atom, down the group basicity of the atom decreases when the size of the atom increases and the electron density decreases.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
